LifeVest: A Precarious and Unproven Bridge . . . to Somewhere
By John Mandrola
August 05, 2013
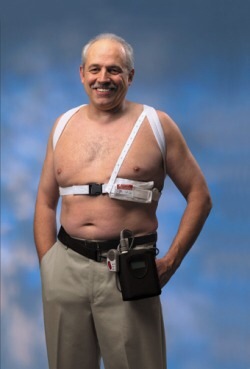
You have to give the marketing folks at Zoll credit. They came up with a nifty name for the wearable cardiac defibrillator (WCD). The LifeVest conjures up images of those fluffy yellow and red jackets we wear on boats and tots wear in pools. So effective has been the marketing that University of Pennsylvania cardiologist Dr Ralph Verdino made use of the comparison in the pages of theJournal of the American College of Cardiology in this 2010 editorial.
Yes, it is indeed true; both safety devices might just preserve a human life. The glaring difference, though, is that the only adverse effect of a floaty is an uneven tan. It’s a different story when one straps a fellow human to a beeping, vibrating, high-voltage vest. For when we do that, we do a lot.
Medicine has grown so fierce, so furious, so ripe with monitors and interventions. Do we also consider that the human organs we aim to monitor and treat come attached to a human mind? A mind that feels intangible things, like fear? Gosh, do I fear the creation of fear. Life comes with so much of that already.
Okay. Okay. Sorry. Let’s crunch some numbers. Then you decide. No, check that: you can help your patient decide.
Scientific data on the LifeVest:
First, when evaluating whether a therapy is effective, it makes sense to start with the randomized controlled clinical trial. You know, the mechanism by which a physician can compare a new treatment with standard care.
Hmm, now we have a problem.
Approved for use for more than a decade, the LifeVest can boast not a single shred of clinical-trial evidence that shackling humans to a beeping, vibrating, shocking vest is any better than standard medical therapy. (The VEST trial is actively recruiting patients, after a nudge from the Centers for Medicare & Medicaid Services in 2011, but 11 years after approval one would have expected at least one trial result.)
If these words sound too much like hyperbole, consider how sure we were when beginning the DINAMIT and IRIS trials. Post-MI patients with high-risk features harbor a high incidence of sudden death, so it was assumed a prophylactic ICD implanted early would affect survival. Not so much. Not even close. Overall mortality, the only kind that counts, was no different with the ICD. Assuming the LifeVest to be beneficial, without any evidence, because it makes sense, reeks of the all-too-typical hubris of American medicine.
Without a clinical trial to evaluate the LifeVest, we must turn to reports culled from manufacturer-controlled databases. Dr Andy Epstein (University of Pennsylvania, Philadelphia) and colleagues reported the most recent and largest of these in theJournal of the American College of Cardiology last week. (Steve Stiles has the summary here .)
In a six-year span, 8453 post-MI patients wore the WCD. Similar to prior database reports, the incidence of shocks was 1.6%. This translates to 8320 patients who received no therapy, while 133 patients received at least one appropriate shock. Survival after the initial shock was 91%, but longer-term survival was lower. Of the 121 patients who survived the initial shock, three died within days and 41 died in the coming months. That leaves 77 patients of the original 8453 who sustained an appropriate shock and were alive. My back-of-the-envelope calculation estimates that we strap the vest on 109 patients for everyone who is alive after a shock. (Of course, all agree that a shock for ventricular arrhythmia does not equate to a life saved.)
Inappropriate shocks were not uncommon and occurred at nearly equal rates (per patient) as appropriate shocks: 99 patients received 114 inappropriate shocks. The authors downplay the shocks, saying there were “no sequelae beyond the immediate pain.” They further minimize the risk (of wide-awake jolts) through the use of numbers rather than words. Here they quantify the chance of an inappropriate shock by monthly risk rather than overall risk. A paltry rate of 0.006 inappropriate shocks per month makes it seem a lot less than the 1.6% overall shock rate for ventricular arrhythmia.
There are no mentions of psychological parameters, sleep disorders, or measures of quality of life. In fact, few of the LifeVest databases report such data. One wonders about the effects of not being able to take a shower without a chaperone and being awakened at night with alarms that might herald a shock?
There are other database studies (here and here). They each report similar statistics, plus these notable findings: as expected, in patients at greater risk for ventricular arrhythmia (resuscitated cardiac arrest, for example), shock rates are slightly higher. At HRS 2013, the WEARIT-II registry noted that 40% of patients who wore the LifeVest went on to improve LV function and no longer met criteria for ICD implantation.
Summary
There are patients at risk of sudden death in whom a permanent ICD cannot be used. Clinical scenarios include patients with the following: high-risk features in the first 40 days after MI, infected ICDs awaiting another device, resuscitated cardiac arrest with slow organ recovery, transient arrhythmogenic disease, like myocarditis, or newly diagnosed nonischemic cardiomyopathy, and suspected diagnosis of an inherited disorder awaiting confirmation of risk.
For many of these patients, a bridge to somewhere seems like a good idea. It makes sense. LifeVest could be that bridge. The emphasis here however falls squarely on the modal verb could. We simply do not know how the LifeVest would compare with optimal medical therapy. We don’t know because there’s no clinical trial. There has been no reason for the company to study such a well-reimbursed product, one that has markedly increased in use of late.
And why has use of the LifeVest taken off? Perhaps it is just enforcement of evidenced-based waiting periods; but perhaps it is also because the device (and its marketing) so elegantly appeal to the most compelling of human instincts—fear of death.
The databases teach us that this bridge to somewhere often goes unused. Most humans strapped with the LifeVest (98%) do not use it, and up to a third of those who do die anyway.
But the circle continues: No caregiver wants his or her patient to die needlessly. And we all agree that a “not-unimportant” fraction of patients have their life preserved by the vest.
Will you excuse me if I use the word conundrum?
So what is left to conclude?
During a spirited (after-hours) discussion at this year’s Heart Rhythm Society sessions, one in which I was expressing negative biases about the LifeVest, a former mentor admonished me, “John . . . if you don’t like how a therapy is being used, don’t blame the therapy, blame the folks who are using it unwisely.”
He is right. I know the LifeVest offers rigorously selected, highly motivated, and intensely educated patients a valuable safety net and a possible bridge to somewhere.
I am also sure no patient needs a LifeVest. And surely no patient should have one without a thorough evaluation and strong confirmation of whether being shackled to a scientifically unproven, beeping, vibrating, shocking vest aligns with their goals.
As fellow human beings, we owe our at-risk patients a candid and truthful discussion.
It gets really tough when the freedom that makes human life so beautiful comes to the fore. It’s a struggle to know how much bubble wrap is too much.
—JMM
theheart.org on Medscape > Trials and Fibrillations with Dr John Mandrola

Leave a comment